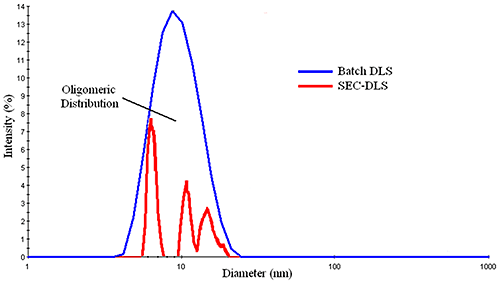
Effectively screening a wide range of formulations in order to select the best candidates for long-term stability studies is a major challenge.”ĭynamic light scattering (DLS) sensitivity has rather been known to start at less than half a nanometer, notes Dan Some, principal scientist at Wyatt. “This process reduces the risk of not finding a suitably stable formulation and consequently delaying commercialization. “Ensuring that the proteins are produced in the proper form, and that the form remains stable all the way from production to the patient, is required,” he states.īecause a stability study involves setting aside quantities of candidate formulations for long periods of time to evaluate shelf life, it becomes essential to minimize the number of candidates and pre-optimize the selection as much as possible, adds Dan Some, principal scientist at Wyatt. It is thus imperative that proteins be reconstituted and/or diluted with a diluent in an ionic environment in which the protein is stable. Proteins are, in addition, particularly sensitive to temperature changes, pH, and conductivity, he says. Proteins are more fragile than typical small-molecule therapeutics, and their function depends not only on their chemical identity (primary structure), but on the shape of the molecule (secondary and tertiary structures), and even multi-subunit proteins (quaternary structure). “A combination of analytical techniques is often needed to better understand or pinpoint the likely cause of instability and identify potential remedies,” she states.Īnother challenge with protein therapeutics is that they are quite complex molecules, remarks John Bak, principal scientist, at PPD Laboratories GMP Lab. Hanna Jankevics-Jones also notes that many analytical techniques used in stability studies measure the secondary effects of instability, such as aggregation, rather than the primary cause of instability, such as reduced repulsion due to increased ionic concentration in the buffer. “If the stresses are significant enough to cause unfolding, this often manifests itself in secondary effects, such as protein aggregation,” she adds. These stresses can reduce or increase the strength of these non-covalent bonds (depending on the magnitude and direction of change) and thereby affect the likelihood of the protein maintaining its structure and stability, she explains. 
A change in the local environment around the protein, such as change in pH, ionic strength, or temperature, or an external factor for example, agitation, can cause stress that affects the non-covalent bonds in the protein’s structure. The technique can also be a reliable method for characterizing protein stability (1).Īmong the most challenging aspects of conducting analytical stability studies on protein therapeutics is protein instability, which can be caused by many different mechanisms, says Hanna Jankevics-Jones, PhD, segment manager, pharmaceuticals, at Malvern Panalytical. Dynamic light scattering (DLS) is a well-known technique for determining sample interactions, particle size, and aggregation of molecules dispersed or dissolved in solution.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
